Levofloxacin in the Treatment of Community-Acquired Pneumonia

MD, Medical Director; Respiratory
Therapy, Spartanburg Regional
Medical Center, Spartanburg, SC, USA.
Fluoroquinolones continue to become an increasingly important class of drugs for treating a wide range of infections, with ofloxacin being one of the most commonly used of these compounds (1). Levofloxacin, the l-isomer of the racemic ofloxacin, is twice as potent as its parent compound, and therefore possesses all of the advantages offered by ofloxacin, as well as providing additional benefits. Improved effectiveness without any increase in side effects makes levofloxacin a very useful and advantageous antibiotic. A favorable pharmacokinetic profile allowing very effective once-daily oral dosing, with an associated increase in compliance, and improved cost-effectiveness make levofloxacin an extremely attractive therapeutic choice in times of increasing cost-containment.
The use of fluoroquinolones in community-acquired pneumonia (CAP) has been one area which has been debated since their introduction, but now with changes in resistance profiles, and the recognition of the role of atypical pathogens it has become clear that they are useful in this setting. To investigate the optimum use of levofloxacin in treating CAP, Penetration interviewed Dr. Charles M. Fogarty, Medical Director; Respiratory Therapy, Spartanburg Regional Medical Center, Spartanburg, South Carolina, USA, who presented the latest results from a multi-center study investigating the safety and efficacy of levofloxacin in this setting.
![]()
Questions
Q4. Could you comment on the use of levofloxacin against atypical pathogens?
Q7. Would you foresee that oral levofloxacin can be used in place of other injectable antibacterials?
Q8. Are there any situations in which you would use levofloxacin in combination with another drug?
Q9. Could you describe the study you performed using levofloxacin in CAP?
Q10. What were the results from the study?
Q11. Were there any adverse effects associated with levofloxacin in the study?
Q12. What is the main benefit of having a once-daily schedule in CAP patients?
Q13. Did you do any cost-analysis in this study?
Q14. Could you comment on resistance in this setting?
Q15. What role do you see for levofloxacin in the immunocompromised patients?
Q17. In the patients that you treat as outpatients do you see problems with compliance?
Q18. What do you see as the areas of future research?
![]()
Answers
Q1. Could you describe the pharmacokinetic features of levofloxacin which you see as being advantageous in the treatment of community-acquired pneumonia (CAP)?
A1. Levofloxacin, which is the active l-isomer of ofloxacin, is twice as potent as its parent compound. Levofloxacin is 100% bioavailable and rapidly penetrates into tissues, where it achieves high levels (2). In fact the plasma levels themselves are bactericidal for the vast majority of community-acquired pathogens, specifically Streptococcus pneumoniae. Other very favourable pharmacokinetic effects include an 80-85% renal excretion with virtually no metabolites, a plasma half-life of six to seven hours and a two to three hour post antibiotic effect allowing the physician to maintain therapeutic levels with once-daily dosing (Table 1).
Table 1. Pharmacokinetics of levofloxacin
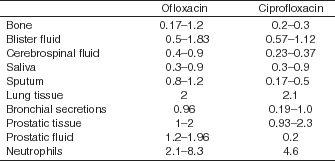
With the potential for bacteremia and the risk of spread to other organs, specifically meningitis, it is reassuring to know that the levels of levofloxacin in the central nervous system (CNS) and other organ systems are higher than with other fluoroquinolones such as ciprofloxacin (Table 2).
Table 2. Tissue penetration of ofloxacin and ciprofloxacin
Q2. The treatment of CAP with fluoroquinolones has been debated. What is your view on using quinolones in this situation?
A2. It is ironic that only a year ago it was assumed that there was no role for quinolones in the treatment of CAP. However this has changed in the last year, with increasing awareness of atypical pathogens, increased antibiotic resistance, increasing numbers of immunosuppressed patients and increasing cost-containment pressure (3-5). All of these factors have led to a reexamination of the role of quinolones in CAP, and certainly levofloxacin should have a major role in this setting.
Q3. What is the antibacterial activity of levofloxacin, and how does this relate to the pathogens commonly encountered in CAP?
A3. S. pneumoniae accounts for 20% of the CAP pathogens in most studies. There is another 30% which is unspecified and probably half of that is S. pneumoniae. Haemophilus and Moraxella are also important pathogens. While ciprofloxacin has given ofloxacin “guilt by association” in the therapy of S. pneumoniae, the fact is that ofloxacin has proven efficacious even in patients with bacteremia, some of whom were only treated with oral therapy (6, 7). Levofloxacin has MIC values of 1.9 for S. pneumoniae, 0.2 for Haemophilus influenzae, and 0.09 for Moraxella catarrhalis. Oral therapy with a once-daily 500 mg dose of levofloxacin produces plasma levels that are well above the MICs for the predominant pathogens, which compares very favorably with the other available antibiotics.
Q4. Could you comment on the use of levofloxacin against atypical pathogens?
A4. We are underestimating the morbidity and mortality of what we used to call “walking” pneumonia (8, 9). By definition, mortality for outpatients is low, but a recent meta-analysis of 33,000 patients showed that mortality for inpatients was 9.8% for Chlamydia and 13.8% for Legionella. Even Mycoplasma, which we would expect to be benign, had a 1.5% mortality rate. Complicating this is the old notion that atypical pathogens can be reliably differentiated from typical pathogens using clinical and radiological findings. Unfortunately, it has been shown that such differentiation is not reliable. In a recent study of 56 patients with Legionella, 41% had a simultaneously positive culture for S. pneumoniae. Clinicians could falsely conclude that a patient with a positive S. pneumoniae culture is doing poorly due to “bad luck” when in fact he has a concomitant, undiagnosed, untreatedLegionella infection. The implication is inescapable: At least in the moderately to severely ill patient, the physician has to provide initial routine coverage for both atypical and typical pathogens. Ofloxacin has been shown in studies to be as good or better than erythromycin in treating the atypical pathogens (1), therefore levofloxacin should be even better. Levofloxacin’s MICs forChlamydia and Mycoplasma are in the 0.5 mg range, with even lower values for Legionella, making it an excellent initial choice.
Q5. Could you summarize what you see as the advantages levofloxacin has over other antimicrobials, including other fluoroquinolones?
A5. Levofloxacin has a very broad spectrum of activity, excellent bioavailability, low toxicity, minimal risk of drug-drug interactions and very importantly, fewer problems with resistance (Table 3). The frequency of one step mutations to resistant organisms appears to be lower with levofloxacin than for other fluoroquinolones. Like the quinolones in general, levofloxacin inhibits DNA gyrase but unlike many of the other quinolones, levofloxacin uses two separate mechanisms to avoid the development of resistance. Another important levofloxacin advantage, is no interaction with drugs such as theophylline (in contrast to other fluoroquinolones, particularly ciprofloxacin). Levofloxacin has a very low side effect profile and although phototoxicity is a theoretical concern, it is definitely less of a problem than with other quinolones such as lomefloxacin and sparfloxacin.
Table 3. Pharmacokinetics of levofloxacin drug interactions
Q6. Intravenous and oral forms of levofloxacin have similar pharmacodynamic properties. How do you use these two administration schedules?
A6. The great thing about levofloxacin is that most of the time one pill can replace a whole intravenous (IV) bag. Oral and IV peak levels are essentially identical and the only difference is the time to maximal concentration. Unless the patient is in shock, unconscious or uncooperative, he can probably be treated with an oral dose. So, even a very sick patient who needs to be in the hospital, even in intensive care, could possibly be treated with oral levofloxacin. One caveat is that many patients are routinely on antacids or H2 blockers. Like all other fluoroquinolones, levofloxacin chelates with antacids, but this problem can simply be avoided by giving the medications two hours apart.
Q7. Would you foresee that oral levofloxacin can be used in place of other injectable antibacterials?
A7. Absolutely. In the United States and throughout the world we are becoming more cost conscious and even though a patient may require hospitalization, he may not need IV therapy. One levofloxacin 500 mg pill may provide broader coverage than most b.i.d. or t.i.d. IV therapy cephalosporins.
Q8. Are there any situations in which you would use levofloxacin in combination with another drug?
A8. If I was worried about resistant staphylococci, the combination of levofloxacin and rifampin would be of interest. Another possibility is to use levofloxacin, which acts mainly via concentration dependent killing and has a two to three hour post antibiotic effect (PAE), and combine that with a cephalosporin or ß-lactam, which generally works in a time-dependent killing manner. By combining drugs from two different classes you should be able to achieve a very broad spectrum of potent activity for sick patients, as well as minimize drug resistance. While the current ATS guidelines for empiric treatment of CAP in hospital patients suggest the combination of a second or third generation cephalosporin and a macrolide it may be better to use levofloxacin instead of erythromycin, particularly as erythromycin resistance continues to develop.
Q9. Could you describe the study you performed using levofloxacin in CAP?
A9. We assessed the efficacy and safety of 500 mg levofloxacin once-daily as empiric monotherapy in a broad spectrum of patients who presented with signs or symptoms of CAP. All patients had a sputum collection which was processed for culture within 20 minutes of presentation. If they were unable to produce a sputum sample they underwent bronchoscopy with lavage of the affected segment. In addition blood cultures, serology for Chlamydia, Mycoplasma, Legionella and urinary antigen for Legionella were obtained, as well as CBC and chemistry profile. Patients were stratified into mild-moderate or severe illness (defined as hypotension in the absence of volume depletion, altered mental status, respiratory rate of 28- 30, requiring intubation, mechanical ventilation, or bacteremia in pretreatment blood cultures). In general, using APACHE II scoring, those with a score of 14- 15 or higher were judged to have severe disease. The initiation of IV or oral therapy was at the investigators discretion. Efficacy was assessed looking at both eradication of the pathogen as well as clinical response.
Q10. What were the results from the study?
A10. Out of 68 patients enrolled, 26 were classified as severe and 42 as mild or moderate. Forty eight patients were admitted, although only 35 were treated with IV therapy. The average duration of IV therapy was 4.3 days. Thirteen in-patients and all 20 outpatients were treated with oral therapy from the outset. The study protocol required that patients be continued on IV therapy for three days, with a total duration of IV and oral therapy of 7- 14 days. Several very sick patients on initial IV therapy were dramatically better after just one dose but due to the protocol they remained on IV therapy for the three days as specified.
Sixty patients were fully evaluable, of whom 57 or 95% were assessed as cured and 3 or 5% assessed as improved. All of the pathogens were eradicated. Twenty four patients had S. pneumoniae, 16 H. influenzae, 8 Chlamydia, 2 H. parainfluenzae, 3 staphylococci, 2 Klebsiella, 1 psittacosis and 1 Legionella. Seven of the 24 S. pneumoniae had positive blood cultures and two of those patients were subsequently diagnosed as HIV positive. Ten patients (14.7%) had atypical pathogens, all of whom were assessed as cured or improved. There was no pattern on initial clinical examination or chest film which allowed investigators to accurately predict ahead of time who had an atypical or typical pathogen.
Q11. Were there any adverse effects associated with levofloxacin in the study?
A11. None of the serious problems encountered were attributable to levofloxacin. Two patients with myocardial infarctions, both had pre-existing risk factors for coronary artery disease. In terms of less serious side effects, the incidence was very low, generally under 5%. The incidence of side effects such as diarrhea did not increase with longer duration of therapy.
Q12. What is the main benefit of having a once-daily schedule in CAP patients?
A12. Once-daily dosing with the potential for early switch to oral therapy is very advantageous for the physician and nurse trying to compress the benefit of yesteryear’s five to six day hospitalization into the current three to four day “allowable”. Instead of running around hanging IV bags all day the nurse hangs just one bag, or even better, gives one pill. Twenty four hours later with culture results back, the patient receives a second dose PO and potentially goes home the next day. The physician is happy, the nurse is happy, the insurance company is happy, and most important of all, the patient is better.
Q13. Did you do any cost-analysis in this study?
A13. We did not do any cost analysis prospectively, but there was an ongoing pneumonia trial with the hospital assessing costs for pneumonia treatment in general. We were advised by the hospital that the average length of stay of our patients, and a great many of them were patients using levofloxacin in this study, adjusting for severity of illness, was one day less. This corresponded to a saving of at least US$1,000. We will assess cost-effectiveness later, but we know ahead of time that it was certainly efficacious and quite cost advantageous.
Q14. Could you comment on resistance in this setting?
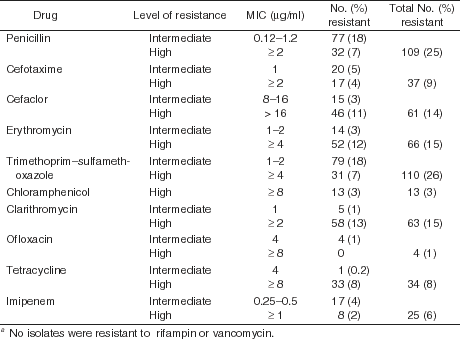
A14. This is a very serious concern with many communities in the United States now hot spots for penicillin-resistant pneumococci. A recent study from Atlanta showed resistance reaching 25% in adults, and even higher in children (Table 4) (10). A third of those strains are multi-resistant to other drugs including erythromycin, sulphamethoxazole- trimethoprim and doxycycline. During 1993- 94 the overall resistance for S. pneumoniae in our center was about 10%. Our laboratory is now reporting an intermediate resistance rate approaching 25%. We are left with the problem that conventional empiric coverage is either going to have to mandate high IV doses of cephalosporins or ß-lactams or alternative drugs such as ofloxacin and levofloxacin. Interestingly in the Atlanta study, ofloxacin has the lowest resistance rate which is part of the reason why we have gone from saying there is no role in CAP for quinolones, to a very real role, particularly for ofloxacin and even more so for levofloxacin.
Table 4. Proportions of pneumococcal isolates resistant to specific antimicrobial drugs from 431 patients in metropolitan Atlanta, January through October 1994a (reference 10)
Q15. What role do you see for levofloxacin in the immunocompromised patients?
A15. An immunocompromised patient is almost totally dependent on the bactericidal activity of the drug administered because his own defenses are impaired by diseases such as alcoholism or HIV infection. The logical therapeutic choice in this setting is a bactericidal drug which has essentially identical MBC and MIC levels. S. pneumoniae is a principal pathogen in this patient population, with one study of HIV patients reporting an incidence of 1% per year, which is five to ten times the usual incidence (11). Levofloxacin in easy to use in HIV patients, as it has not been found to have any significant drug interactions with the antivirals commonly used by these patients. I think we will see more long-term use of drugs like levofloxacin in treating specific infections such as tuberculosis, in HIV and alcoholic patients.
Q16. From the results of your clinical trial and experience could you comment on the use of levofloxacin in general lower respiratory tract infections?
A16. I would use levofloxacin as initial therapy in patients who are moderately to severely ill, although they may not all require hospital admission. I would use it in patients where I can not afford to be wrong or to lose a few days of treatment; this would include chronic obstructive pulmonary disease (COPD) patients with good Gram stain evidence of an acute infection.
Q17. In the patients that you treat as outpatients do you see problems with compliance?
A17. Absolutely. The minute a patient starts to feel better the likelihood of his continuing to take pills two to three times a day decreases dramatically. Therefore a once a day drug such as levofloxacin that is bactericidal, concentration dependent in its killing and acts quickly up front offers a very important advantage.
Q18. What do you see as the areas of future research?
A18. Studies investigating efficacy and safety of once-daily higher doses of levofloxacin such as 750 mg. Also the concomitant use of a second antibiotic, looking for synergy and lower rates of resistance. The problem is that the second drug would have to be potent in an oral form, which poses some interesting challenges for a lot of the second drugs we would use.
![]()
References
- Krop LC. Ofloxacin: why the increased popularity? J Pharm Technol 1996; 12: 88- 95.
- Davis R, Bryson HM. Levofloxacin: a review of its antibacterial activity, pharmacokinetics and therapeutic efficacy. Drugs 1994; 47: 677- 700.
- Niederman MS, Bass JB, Campbell GD, Fein AM, Grossman RF, Mandell LA, et al. 1993. Guidelines for the initial management of adults with community-acquired pneumonia: diagnosis, assessment of severity, and initial antimicrobial therapy. Am Rev Respir Dis 1993; 148: 1418- 26.
- Bartlett JG, Mundy LM. Community-acquired pneumonia. N Engl J Med 1995; 333: 1618- 24.
- Fine MJ, Smith MA, Carson CA, Mutha SS, Sankey SS, Weissfeld LA, et al. Prognosis and outcomes of patients with community-acquired pneumonia. JAMA 1996; 275: 134- 41.
- Gentry L, Lipsky B, Farber MO, Tucker B, Rodriguez-Gomez G. Oral ofloxacin therapy for lower respiratory tract infection. South Med J 1992; 85: 14- 7.
- Gentry L, Rodriguez-Gomez G, Kohler R, Khan F, Rytel M. Parenteral followed by oral ofloxacin for nosocomial pneumonia and community-acquired pneumonia requiring hospitalization. Am Rev Respir Dis 1992; 145: 31- 5.
- Kauppinen MT, Lahde S, Syrjala H. Clinical picture of community-acquired pneumonia requiring hospital treatment: a comparison between chlamydial and pneumococcal pneumonia. Thorax 1996; 51: 185- 9.
- Lieberman D, Porath A, Schlaeffer F, Boldur I. Legionellaspecies community-acquired pneumonia. a review of 56 hospitalized adult patients. Chest 1996; 109: 1243- 9.
- Hofmann J, Martin C, Farley M, Baughman W, Facklam R, Elliott J, et al. The prevalence of drug-resistant Streptococcus pneumoniae in Atlanta. N Engl J Med 1995; 333: 481- 6.
- Hirschtick RE, Glassroth J, Jordan MC, Wilcosky TC, Wallace JM, Kvale PA, et al. Bacterial pneumonia in persons infected with the human immunodeficiency virus. N Engl J Med 1995; 333: 845- 51.